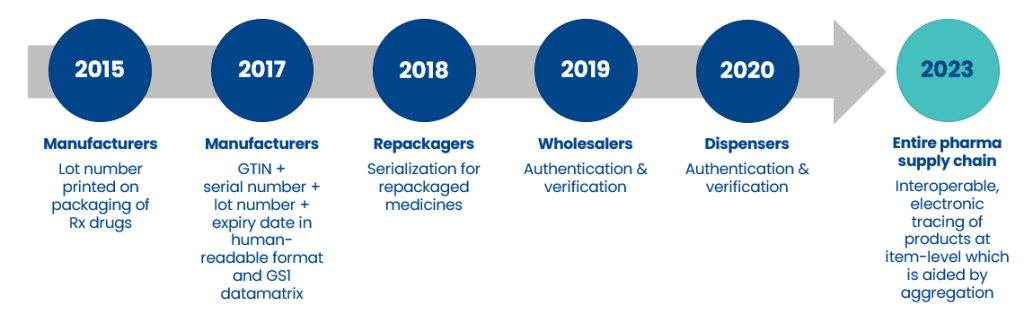
Regulations put in place by the US Drug Supply Chain Security Act (DSCSA) and the EU Falsified Medicines Directive (FMD) have significantly progressed the security of the pharmaceutical supply chain to better protect patients against the threat of counterfeit, falsified, contaminated, or otherwise harmful pharmaceutical products.
As companies worked towards implementing serialization to meet the regulatory deadlines, a regular topic of conversation was whether to introduce aggregation capabilities alongside serialization.
While many companies have been working toward implementing aggregation to support the upcoming DSCSA 2023 deadline, there are still mixed opinions as to whether investing in aggregation will deliver worthwhile returns in regulatory markets where that is not mandated.
In this article Alexander Klemp, Senior Technical Services Project Manager at Sharp Packaging describes the business rationale for including aggregation as part of your serialization strategy, beyond the regulatory requirement.
What are Serialization and Aggregation?
Serialization is the creation and assignment of a unique serial number to each individual unit of a pharmaceutical product. This number allows that unit to be traced on its journey through the supply chain and, when scanned, the serial number will provide all the unique data associated with that unit such as its point of origin, batch number, and expiration date.
Aggregation refers to the process whereby a relationship is established between each level of the packaging hierarchy, from the individual unit level to shipper case to pallet. A unique serial number is created at each level, which allows pharma companies to identify and authenticate products more quickly and easily as they move through the supply chain. This hierarchical relationship between lower packaging levels, a single bottle containing drug product, for example, and higher levels, a shipper case carrying multiple bottles, means the barcode on the pallet can be scanned to obtain information about each packaging level within the batch down to unit level, including bulk drug product lot details.
The Drug Supply Chain Security Act 2023
In November 2018, the regulation making item-level serialization mandatory came into force. Since this deadline, all manufacturers have been required to print lot number, expiry date, and serial number on the packaging of their prescription drugs. Having now complied with the FDA requirement the industry is recognizing the value and potential return on investment of the serialized supply chain, including aggregation.
The final DSCSA deadline in 2023 will see legislation change again with the Enhanced Drug Distribution Security (EDDS) phase intended to further enhance drug distribution safety, security, and efficiency. The regulation requires interoperable, electronic tracing of products at the package level, which in turn generated a business requirement for aggregation in order to support data exchange for every product transaction.
There are many ways to comply with this new regulation. However, the most adopted approach is aggregation upon initial deployment of serialization. This approach means that manufacturers deploy only one set of changes to their products and are ready well in advance of the 2023 deadline.
Aggregation: How it Works
With aggregation, unit packages are serialized with unique numbers, placed into shipper cases containing their own unique serial numbers on affixed serialized labels, and placed onto similarly serialized pallets. This establishes a parent-child hierarchy which allows each product to be traced more precisely in the supply chain.
Why Aggregate Now?
Implementing aggregation as part of your serialization strategy is worth giving serious consideration.
Operational Efficiency and Enhanced Quality Management
There is a misconception that aggregation can slow down the packaging process. While it is true that adding process steps to account for aggregation can reduce the throughput of a packaging line, what often isn’t considered is that there is less time spent identifying, locating, and correcting a deviation during quality checks on a packaging line or after a lot has been released. Aggregation offers additional tools for investigating issues within a packaging or bulk drug product lot, thereby providing supply chain optimization as well as enhancing quality management.
Because of the systematic requirement that aggregation brings, specific units must be packed into a specific shipper case, thus dictating packing order and removing any randomness. If the process is not followed, it is very evident and easy to trace and correct the issue before the lot is closed.
Aggregation makes the verification of final lot count at the close of packaging activities more efficient because it quickly confirms the final counts ensuring the physical packaging lot matches the electronic data. Additionally, the ability to electronically track detailed information during the packaging process removes the need to open packaged shipper cases to confirm and verify contents if discrepancies do arise.
For product recalls, the positive impact is even greater when the product has already been sent for distribution. Aggregation timestamps can be used in addition to the packaging batch record to help isolate the point in time where a specific event happened, for example, the introduction of a new bulk lot in the case of master lotting. This allows for the recall of specific units within the packaged lot to be located and securely removed from the supply chain allowing the remaining product to continue onto market.
Implementation Pressures
While aggregation in the US is not explicitly a regulatory requirement, it is ultimately driven by the business to meet EDDS within the DSCSA. Building in aggregation from the outset means there will be no need to retrofit it to the serialization program in the future. Consider the original 2018 DSCSA deadline, many companies decided to aggregate right away to avoid the need to make any changes in the future and risk further disruption to operations and supply. While there was certainly hesitation at first, many companies quickly saw the value in this approach. In order to save time, it makes more sense to deploy changes all at once, rather than in phases. There have also been multiple other regulatory rollouts in countries with similar mandates to the US DSCSA with earlier deadlines, for example, Russia.
Implementing serialization with aggregation can be complex, but it doesn’t have to be. The first thing to consider is the physical hardware required from a capital expenditure perspective, the cost can be high and equipment lead times can cause delays. Equally, the software required to generate and manage serial numbers on a packaging line as well as connectivity to the data cloud will involve careful planning and collaboration with Electronic Product Code Information Services (EPCIS) vendors who will advise on data management and security.
Working with an experienced packaging partner can minimize the challenges. Service providers who specialize in pharmaceutical packaging services will be able to provide a deeper perspective, giving their clients a granular breakdown of the process. The quality and quantity of data collected and reported to their clients can also be a powerful tool offering a wealth of information and insight. In turn, this means that contract packagers are not only well placed to offer a minimum of three levels of aggregation but also able to manage the associated data efficiently and securely.
Looking Ahead
New security regulations will inevitably continue to come into force around the world in an effort to continue to protect all stakeholders against counterfeiting and diversion. Whether mandated or not, aggregation can, in the long run, offer pharma companies additional unforeseen benefits such as greater traceability, visibility, and control in the management of their supply chain.